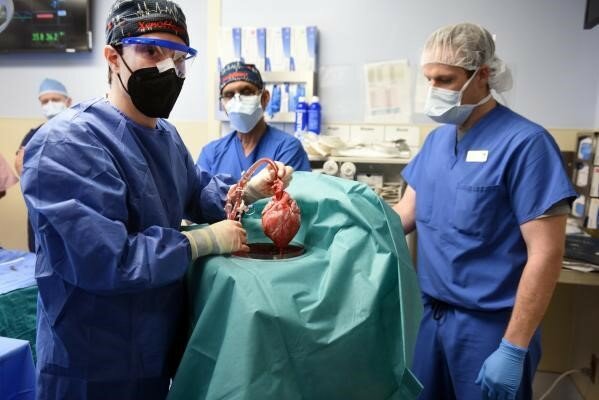
The idea of accepting an animal organ in place of our own might sound repugnant at first. Is it a real danger to our human identity? In a “world-first” operation, a genetically modified pig’s heart was transplanted into a human.
The operation was Mr. Bennett’s last resort, a shot in the dark that could save his life. After physicians at the University of Maryland Medical Center declared that Mr. Bennett’s body was unsuitable for a human donor transplant or an artificial heart pump, a new option had to be discovered.
Despite the surgery being considered a major scientific breakthrough, the concept of xenotransplantation is not novel. It typically refers to any procedure involving implantation, transplantation, or infusion of cells, tissue, or organs from an animal source to a human.
Theoretically, animals that mostly resemble humans genetically would make potential donors. Hence, non-human primates were extensively studied and explored for xenotransplantation during the 20th century. Other medical procedures also exist. Each is defined by the organ being transplanted, the nature of the donor, and the recipient. For example, an refers to the transplant of a tissue or organ within the same species such as from a donor human to a recipient human or sheep to sheep.
This article aims to present the obstacles that arose along the way of using pigs’ organs in xenografts to save humans as well as the repercussions of such a breakthrough to the field of medicine.
“It creates the pulse, it creates the pressure, it is his heart,” explained Dr. Bartley Griffith, the director of the cardiac transplant program at the medical center. Mr. Bennett was diagnosed with terminal heart disease and wasn’t eligible for a human heart transplant. The surgery was his last hope of surviving. Currently, more than 106,000 men, women, and children are registered on the national transplant waiting list as by OrganDonor.gov. Statistics show that more than a dozen people die each day on the waiting list. Worse, your chance of receiving a kidney transplant is as low as 25 percent if you are on the waiting list. In the light of the acute shortage of organs donation, the animal-to-human transplant was seen as a great opportunity to save more lives.
In 2021, kidneys were from genetically modified pigs to two legally brain-dead people with success. As expected, the kidneys functioned properly in their new hosts who were sustained on ventilators. However, Mr. Bennett is the first human who had the chance to survive and recover after transplantation of a genetically modified pig’s organ. This watershed moment offers a glimpse of hope for thousands in need of organs.
The transplant, however, raised many concerns over ethical issues, breeding difficulties, and transmission of infections across species. Activists claim that animals have the right to live without having their genes manipulated and all the suffering it entails to the animal. Also, the extensive monitoring of animals for transplant purposes is crucial to avoid transmissible diseases.
Most importantly, scientists have feared the risk of triggering a dangerous immune response against the newly transplanted organ. However, physicians have regulated all genetic players that would lead to undesired results. The immunobiology behind successful transplant operations relies mainly on the Major Histocompatibility Complex (MHC). In humans, the MHC is known as the Human Leukocyte Antigen (HLA), which is a complex set of genes that encode proteins that help the immune system identify foreign bodies. They are present on the surface of all cells and mark them as “self” to not trigger any immune response. Every human has a different set of HLA proteins based on their genetic package. As such, graft rejections occur when the immune system of the recipient (the T cells are central in the rejection of grafts) destroys the foreign cells, tissues, or organs displaying different HLA proteins rendering them as “non-self.”
On one hand, a Xenograft would elicit an immune response because of the different species at play and the difference in the MHC protein among animals and humans. On the other hand, an autograft, which consists of transplantation of tissues or organs within the same individual, is the least likely to elicit an immune response.
Scientists have used the genome-editing technique called to edit ten genes in the pig. The technique offers scientists the chance to knock in or introduce genes and knock out or inactivate other genes. In our case, three genes in the pig were identified to be responsible for rapid antibody-mediated rejection of pigs’ organs by humans. These genes were knocked in the pig’s genome. Also, six human genes that are responsible for the human immune system to accept the pig’s heart were knocked in. Finally, to prevent the excessive growth of the pig’s heart tissue in the recipient’s body, scientists have identified the gene responsible for it and knocked it out from the pig’s genome.
Developing genetically modified pigs is a long rocky road. For two decades, David Ayares, who is Revivicor’s executive vice president, has worked on developing pigs suitable for human transplant surgeries. “We’ve marched down this road over the last decade, going from a one-gene pig to a two- to a five- to an eight to a 10, and we rationalized each of those and made modifications along the way,” said Ayares.
Along with genetic modifications to the pig, the surgery team has used immunosuppressive drugs designed to suppress Mr. Bennett’s immune system, hence preventing the body from rejecting the organ. At 57, Mr. Bennett is doing well after the seven-hour procedure in Baltimore. Besides, the breakthrough has renewed the debate over the ethicality of using animals for organs. Historically, pigs are always chosen as organ donors thanks to their close physiological features. Pigs have human-sized organs and short gestation periods.
Animal activists reject these arguments advocating for the protection of animals at all costs.
The business of engineered animals used in xenografts is booming. Many companies are producing pigs that do not transmit retroviruses or other diseases. Other companies in New Zealand are breeding small-sized pigs whose kidneys do not outgrow human-sized kidneys without growth hormone manipulation.
Another promising industry that satisfies animal activists is the artificial organ industry. Advances in 3D printing and tissue engineering would create made of blood and flesh.